
As such, cerium oxide is a material of interest for solid oxide fuel cells (SOFCs) in comparison to zirconium oxide. Energy & fuels ĭue to the significant ionic and electronic conduction of cerium oxide, it is well suited to be used as a mixed conductor. The glowing element is mainly ThO 2 doped with CeO 2, heated by the Ce-catalyzed oxidation of the natural gas with air.īuilding on its distinct surface interactions, ceria finds further use as a sensor in catalytic converters in automotive applications, controlling the air-exhaust ratio to reduce NO x and carbon monoxide emissions. 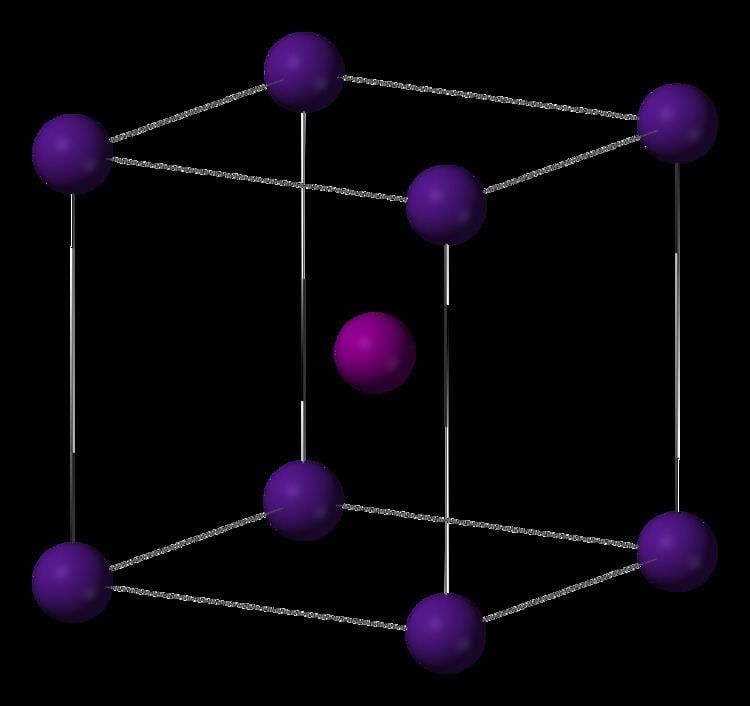
A glowing Coleman white gas lantern mantle. Another small scale but famous example is its role in oxidation of natural gas in gas mantles. One small but illustrative use is its use in the walls of self-cleaning ovens as a hydrocarbon oxidation catalyst during the high-temperature cleaning process. The interconvertibility of CeO x materials is the basis of the use of ceria for an oxidation catalyst. Its reduced derivative Ce2O3 reduces water, with release of hydrogen. It catalyses the water-gas shift reaction. Other niche and emerging applications Catalysis ĬeO 2 has attracted much attention in the area of heterogeneous catalysis. It function by converting green-tinted ferrous impurities to nearly colorless ferric oxides. 
In its other main application, CeO 2 is used to decolorize glass. For hobbyists, it is also known as "opticians' rouge". For this purpose, it has displaced many other oxides that were previously used, such as iron oxide and zirconia. The principal industrial application of ceria is for polishing, especially chemical-mechanical planarization (CMP). Applications Ĭerium has two main applications, which are listed below. Occurrence of cerianite-(Ce) is related to some examples of cerium anomaly, where Ce - which is oxidized easily - is separated from other REEs that remain trivalent and thus fit to structures of other minerals than cerianite-(Ce). It is often found in names of minerals bearing rare earth elements (REEs). The "-(Ce)" suffix is known as Levinson modifier and is used to show which element dominates in a particular site in the structure. It is a rare example of tetravalent cerium mineral, the other examples being stetindite-(Ce) and dyrnaesite-(La). Natural occurrence Ĭerium(IV) oxide occurs naturally as the mineral cerianite-(Ce). Controlling such surface interactions is key to harnessing ceria in catalytic applications. The presence of oxygen vacancies at terminating ceria planes governs the energetics of ceria interactions with adsorbate molecules, and its wettability. The diffusion rate increases as the defect concentration increases. Since the oxygen atoms in a ceria crystal occur in planes, diffusion of these anions is facile. Undoped and doped ceria also exhibit high electronic conductivity at low partial pressures of oxygen due to reduction of the cerium ion leading to the formation of small polarons. These factors give ceria favorable performance in applications as a solid electrolyte in solid-oxide fuel cells. Increasing the concentration of oxygen defects increases the diffusion rate of oxide anions in the lattice as reflected in an increase in ionic conductivity. The primary defects of concern are oxygen vacancies and small polarons (electrons localized on cerium cations). In the most stable fluorite phase of ceria, it exhibits several defects depending on partial pressure of oxygen or stress state of the material. The number of oxygen vacancies is frequently measured by using X-ray photoelectron spectroscopy to compare the ratio of Ce 3+ This material has the formula CeO (2− x), where 0 500 ☌. At high temperatures it releases oxygen to give a non-stoichiometric, anion deficient form that retains the fluorite lattice. Structure and defect behavior Ĭerium oxide adopts the fluorite structure, space group Fm 3m, #225 containing 8-coordinate Ce 4+ and 4-coordinate O 2−. Ĭerium(IV) oxide is formed by the calcination of cerium oxalate or cerium hydroxide.ģ, which is unstable and will oxidize to cerium(IV) oxide. This step exploits the low solubility of CeO 2 and the fact that other rare-earth elements resist oxidation. After extraction of the metal ions into aqueous base, Ce is separated from that mixture by addition of an oxidant followed by adjustment of the pH. Its principal ores bastnaesite and monazite. The distinctive property of this material is its reversible conversion to a non-stoichiometric oxide.Ĭerium occurs naturally as oxides, always as a mixture with other rare-earth elements. It is an important commercial product and an intermediate in the purification of the element from the ores. It is a pale yellow-white powder with the chemical formula CeO 2. Cerium(IV) oxide, also known as ceric oxide, ceric dioxide, ceria, cerium oxide or cerium dioxide, is an oxide of the rare-earth metal cerium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
